22 Nov 2025

A GMP audit in the pharmaceutical sector is a formal evaluation of a facility to determine its compliance with Good Manufacturing Practices (GMP). These audits are conducted to ensure that pharmaceutical drugs are consistently produced and controlled according to quality standards that safeguard their safety, efficacy, and reliability. They play a critical role in confirming that manufacturing processes meet the stringent requirements set by regulatory authorities.
To understand how GMP audits in the pharmaceutical industry works, you have to first dive into what GMP audits constitute in general.
A GMP audit is a systematic and independent evaluation of a pharmaceutical or related manufacturing facility to verify its compliance with current GMP (cGMP) standards. These audits ensure that the manufacturing processes, systems, and controls in place consistently produce products that meet established quality, safety, and efficacy requirements.
Unlike regulatory inspections conducted by health authorities, GMP audits are typically carried out by internal quality teams or external qualified auditors—such as consultants or clients—as part of routine oversight or supplier qualification programs. They are essential for identifying potential gaps in GMP compliance and proactively addressing issues before they impact product quality or trigger regulatory action.
A typical GMP audit involves:
The audit often follows a structured checklist covering critical areas such as:
By regularly conducting GMP audits, manufacturers demonstrate their commitment to maintaining high-quality standards and ensuring patient safety throughout the product lifecycle.
GMP compliance audits play a vital role across industries such as pharmaceuticals, food, cosmetics, and medical devices, where product quality and consumer safety are non-negotiable. These audits are not just regulatory formalities—they are essential checkpoints that verify whether a company’s manufacturing systems, processes, and personnel meet the expected standards of safety, consistency, and control.
Health authorities worldwide, such as the FDA, EMA, and WHO, expect organizations to routinely conduct internal GMP audits as part of self-inspection programs. For instance, the EU GMP Chapter 9 outlines clear expectations for ongoing self-assessments to ensure current compliance with the most updated cGMP guidelines. These audits help organizations proactively identify gaps before they escalate into costly violations, recalls, or regulatory actions.
GMP audits are also a fundamental part of supplier qualification and continuous oversight processes. They ensure that every element of the manufacturing lifecycle—from raw materials and equipment to personnel practices and documentation—is aligned with regulatory requirements and internal SOPs.
Conducting routine GMP audits offers the following benefits across industries:
Whether an audit is announced or unannounced, being audit-ready at all times demonstrates a company’s commitment to quality, transparency, and regulatory excellence.
For the pharmaceutical scenario, the following pivotal factors remain the driving force behind implementing GMP audits:
Conducting a Good Manufacturing Practice (GMP) audit effectively in the pharmaceutical industry involves a well-structured approach that ensures all processes, personnel, and records align with current regulatory expectations. Below are key steps to follow for conducting a successful GMP audit:
Start by reviewing the latest GMP standards and inspection frameworks issued by relevant authorities such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and WHO. Ensure your audit plan aligns with region-specific regulations, including FDA’s 21 CFR Parts 210/211 and EU GMP Annexes. Understanding inspection administrative rules and audit protocols will help the audit team assess operations against accurate benchmarks.
An essential part of any GMP audit is evaluating the company’s CAPA system. Ensure that all previously identified nonconformities have been addressed through documented corrective actions. Additionally, assess whether preventive measures are in place to avoid recurrence of issues. All CAPA activities should be traceable, justified, and backed by objective evidence.
Personnel competence plays a vital role in GMP compliance. All employees, particularly those involved in manufacturing, quality control, and warehouse operations, should be trained regularly on GMP standards, hygiene protocols, deviation handling, and documentation practices. Auditor interviews with staff will assess not just knowledge, but also real-world application of training.
Documentation forms the backbone of GMP audits. Prior to the audit, confirm that all required documents—such as batch production records, quality control data, equipment calibration logs, and cleaning validations—are current, complete, and easily retrievable. Also, verify the accuracy of your manufacturing site registration and product licenses.
Beyond core GMP records, ensure that supporting documents such as SOPs, training logs, change control logs, and maintenance reports are compiled and organized. These records demonstrate the implementation of a quality management system and provide context for operational decisions during the audit.
Evaluate past internal and external audit reports to ensure that all findings have been addressed. Auditors often follow up on previous issues to verify that appropriate resolutions were implemented. This retrospective review helps reinforce a continuous improvement approach.
Performing internal mock audits is a proactive strategy to identify potential gaps and prepare employees for the audit environment. These simulations replicate the actual audit flow, from document reviews to facility walkthroughs and personnel interviews. Mock audits also reduce staff anxiety and help teams practice confident and accurate communication.
A GMP audit checklist for the pharmaceutical industry should be thorough, covering all critical aspects of compliance such as:

This checklist helps ensure that all GMP elements are properly monitored and compliant with regulatory expectations, reducing the risk of quality failures or enforcement actions.
In the context of GMP audits, a nonconformity refers to a failure to meet established regulatory or internal requirements. These deviations can arise from either processes or products, often due to inadequate controls, poor documentation, or failure to follow standard operating procedures (SOPs). During GMP inspections, nonconformities act as key indicators of weaknesses in a company’s quality management system and provide insight into areas requiring corrective and preventive actions (CAPAs).
Below are five of the most common GMP-related nonconformances observed during pharmaceutical inspections:
Lack of Adequate Documentation – Proper documentation is a cornerstone of GMP compliance. A recurring nonconformance in inspections is the absence of complete, accurate, and timely records. Incomplete documentation—whether related to batch production, quality control testing, equipment maintenance, or deviation handling—can severely hinder traceability and regulatory accountability. Inspectors often review records in detail to assess process reliability, and gaps in documentation frequently trigger audit findings.
Inadequate Training of Internal Auditors and Personnel – A lack of sufficient training programs, especially for internal auditors, can lead to unrecognized deficiencies and poor audit outcomes. Inadequately trained personnel are more likely to deviate from SOPs or misinterpret GMP requirements, resulting in preventable nonconformities.
Weak Change Control Management –Change control is critical to managing risks associated with modifications to processes, equipment, or facilities. GMP audits frequently identify issues where changes were implemented without proper risk assessment, approval, or documentation.
Ineffective Supplier and Vendor Management – Failing to qualify or monitor suppliers can result in the procurement of substandard raw materials or services, directly affecting product quality and safety. This often appears in audits as a breakdown in supply chain oversight.
Poor Equipment Maintenance and Calibration – Equipment that is not properly maintained or calibrated poses a major risk of contamination, batch failure, or inaccurate test results. Auditors frequently find nonconformances related to equipment logs, overdue maintenance, or lack of cleaning validation.
Third-party GMP audits offer a fresh, objective perspective that internal teams may lack due to familiarity with daily operations. Over time, personnel often become so accustomed to existing procedures and environments that they may miss subtle signs of non-compliance or inefficiencies. An experienced and specialized third-party audit team, well-versed in applicable GMP standards and manufacturing practices, brings in a wealth of expertise to evaluate your systems critically and constructively.
These auditors don’t just conduct site inspections—they provide actionable insights by identifying overlooked compliance gaps, uncovering operational weaknesses, and highlighting potential risks that internal audits might miss. Their findings can help companies create more robust corrective action plans, refine daily practices, and improve employee training to align better with regulatory expectations. Ultimately, third-party audits enhance product quality, operational efficiency, and overall GMP readiness.
To manage such audits effectively, pharmaceutical companies can benefit from adopting purpose-built digital tools like Smart Audit. As a digital audit management solution, Smart Audit is designed to eliminate the inefficiencies of paper-based or manual audit systems. It streamlines scheduling, execution, and reporting of audits—making it easier to track findings, assign corrective actions, and maintain real-time compliance. With Smart Audit, pharma manufacturers can take full advantage of both internal and third-party audits to continuously raise the standard of their GMP performance.
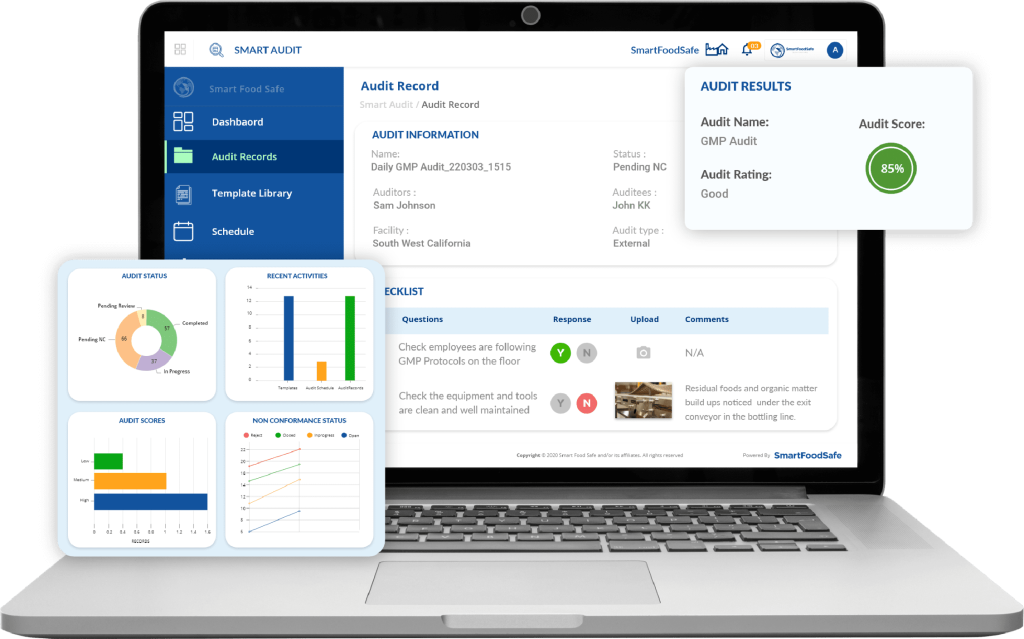
Have a consistent, working internal audit strategy that continuously brings about compliance in the middle of regulatory updates with Smart Audit’s audit management features.
Have a consistent, working internal audit strategy that continuously brings about compliance in the middle of regulatory updates with Smart Audit’s audit management features.

Signup to receive latest news, insights and updates on audits management