10 Dec 2025

FSMA audit day can feel like a stress test for your people, records, and systems. Auditors will methodically trace decisions, logs, and product flows to confirm you’re practicing prevention, not just checking boxes. That pressure is normal, but it’s avoidable. FSMA moved U.S. food safety from a reactive “fix it after a problem” model to a prevention-first, science-based approach, and that shift is exactly what auditors will evaluate. In this guide you’ll get a playbook-style, step-by-step framework for preparing your Food Safety Plan, organizing the evidence auditors want to see, running internal gap assessments and mock recalls, training the response team, and using digital tools to make readiness repeatable and defensible.
FSMA the Food Safety Modernization Act is the U.S. federal framework that re-centers food regulation on preventing foodborne illness rather than reacting to it. That means facilities covered by FSMA must identify hazards, implement risk-based preventive controls, verify those controls work, and keep records that show continuous oversight. Non-compliance can lead to warning letters, import refusals, forced recalls, and significant reputational and legal risk, so audit readiness isn’t just regulatory housekeeping; it’s business protection.
Who it applies to: food manufacturers, processors, certain farms, importers, transporters and distributors, each covered party has specific rules and expectations under FSMA.
Below are the core FSMA rules most likely to shape your audit scope and the documents auditors will request:
Preventive Controls for Human Food (PCHF / PC-Human): Requires a written Food Safety Plan based on hazard analysis, identification of preventive controls, monitoring, verification and corrective actions, typically developed or overseen by a Preventive Controls Qualified Individual (PCQI).
Produce Safety Rule: Science-based standards for growing, harvesting, packing and holding produce, with emphasis on agricultural water, worker hygiene, and equipment sanitation.
Foreign Supplier Verification Program (FSVP): Importers must verify foreign suppliers’ food-safety measures and keep FSVP records. Auditors will inspect FSVP files and verification activities.
Sanitary Transportation Rule: Covers sanitation and temperature controls during transport to prevent contamination.
Mitigation Strategies for Intentional Adulteration (Food Defense): Requires a food defense plan that identifies vulnerable points and mitigation strategies against intentional contamination.
Traceability & Recordkeeping (Food Traceability Rule / KDEs): Certain high-risk foods require additional traceability records (Key Data Elements — KDEs) and mock recalls. Many traceability records must be kept for two years.
Auditors typically cross-check the Food Safety Plan against what actually happens on the floor:
This is the operational heart of the playbook. Each step includes immediate actions, deliverables, and who owns it.

Actions:
Actions:
Actions:
Actions:
Actions:
Actions:
Actions:
These below are the repeat observations auditors make, fix them proactively:
Missing or incomplete monitoring logs: Implement digital time-stamped logging to reduce gaps.
Operators not trained on CCPs or PCIs: Run role-specific, competency-assessed training and keep attendance records.
Outdated hazard analysis or FSP: Schedule annual or process-change reviews and PCQI re-approval.
Poor supplier verification: Keep COAs, supplier audits, and FSVP evidence current.
Inconsistent environmental monitoring: Standardize sampling plans and trend analysis.
Weak food defense plans: Map vulnerabilities and document practical, tested mitigations.
Fixes: automation for logs, scheduled training calendar, supplier scorecards, internal audit cadence, and documented verification.
A FSMA audit might feel intense, but the actual workflow is very structured. Once you understand how the day is organised, it becomes much easier for your team to stay calm, answer confidently, and avoid mistakes.
Below is a detailed, easy-to-understand walkthrough of what a real FSMA audit looks like from start to finish.
The audit begins with a short introduction meeting. This is where the auditor explains:
This meeting is not a test, it’s simply orientation.
What you should provide at this stage
To start smoothly, hand over two things:
Preparing these early sets a professional tone and signals that your team is organized and audit-ready.
After the opening meeting, the auditor dives into the documentation. FSMA is heavily documentation-driven, so this step often takes the most time.
They will examine:
Pro Tip:
Assign a single “document retrieval lead.”
This person is responsible for pulling up files quickly, clearly, and without unnecessary conversation.
Slow or unorganized document retrieval often raises red flags.
After reviewing your documentation, the auditor moves into the facility. This is sometimes the most stressful part because it tests real-world execution, not just paperwork.
The auditor will observe whether:
In simple terms:
They are checking whether what you wrote in your Food Safety Plan is actually happening on the floor.
Common walkthrough hotspots:

Frontline workers play a critical role in the audit. Auditors will often stop operators and ask simple but very important questions, such as:
These questions help auditors confirm:
Coaching Tip for Your Team
Make sure workers understand:
For example :
Bad answer : Well, usually I sanitize, but sometimes if we’re very busy
Good answer : I sanitize the equipment every 4 hours, as trained and as written in the SOP.
At the end of the audit, the auditor will sit with your team again to share:
This meeting is crucial because it clarifies exactly what you need to fix.
What you should do during the closing meeting:
Accurate notes ensure your CAPA plan is aligned with the auditor’s expectations.
Practical Tip: Keep a Running Observation Log Throughout the Audit
Assign someone on your team to quietly track:
This creates a real-time list of likely findings, allowing you to prepare more complete, accurate, and timely corrective actions.
A typical FSMA audit flows like this:
Understanding the flow helps you:
Practical Tip :
Always keep a running log of auditor observations. After the audit, submit a timely, well-documented CAPA plan that includes root cause analysis, corrective action steps, and effectiveness verification.
Digitalisation is one of the fastest ways to shorten auditor time and strengthen evidence
Treat FSMA readiness as a continual program, not a one-off project. A documented, validated Food Safety Plan; reliable monitoring and traceability; a trained audit response team; and the right mix of digital tools will reduce audit time, lower recall risk, and protect your brand. Run regular internal audits, automate evidence capture where possible, and treat every audit observation as an opportunity to strengthen processes and demonstrate continuous improvement. That’s how compliance becomes competitive advantage.
The Food Safety Plan, SOPs/CGMPs, monitoring and verification logs, CAPA records, supplier files/FSVP (if importing), traceability records and training evidence are the core items auditors request.
Annually, and whenever a significant process, supplier or formulation change occurs. PCQI review and revalidation should follow major changes.
HACCP is hazard analysis and critical control point methodology focused on controlling CCPs; FSMA (and PC-Human) includes HACCP-like elements but adds preventive controls, supplier verification, PCQI responsibilities, and broader traceability and recordkeeping expectations.
Many FSMA records (including traceability KDEs) must be kept for 2 years; some records may have different retention periods depending on the rule and record type. Check the specific rule guidance.
Facilities subject to Preventive Controls rules generally need a PCQI — the person who develops or oversees the Food Safety Plan and has training equivalent to the FSPCA curriculum or equivalent experience.
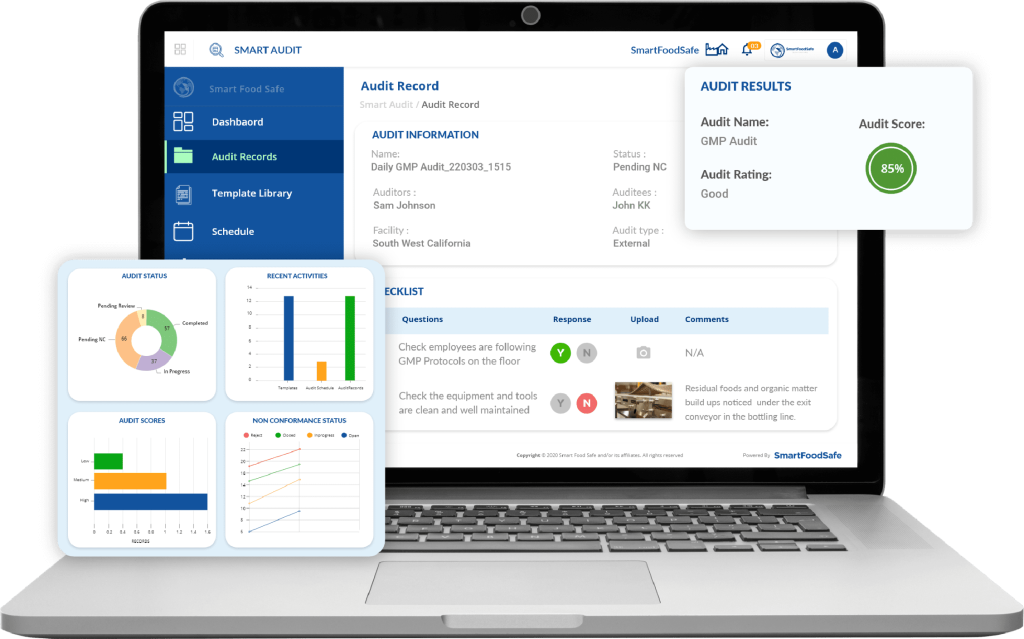
Have a consistent, working internal audit strategy that continuously brings about compliance in the middle of regulatory updates with Smart Audit’s audit management features.
Have a consistent, working internal audit strategy that continuously brings about compliance in the middle of regulatory updates with Smart Audit’s audit management features.

Signup to receive latest news, insights and updates on audits management