16 Jan 2026

Your last FDA inspection didn’t go as smoothly as you’d hoped. The inspector spent an hour digging through your audit files, asking why certain CAPAs weren’t closed on time, and questioning gaps in your documentation. You had the answers, somewhere, but finding them in your maze of spreadsheets and paper trails was like searching for a needle in a haystack.
Sound familiar?
Here’s the reality: paper-based audit management and scattered Excel files aren’t just inefficient anymore, they’re a compliance liability. When regulatory bodies can show up unannounced and expect instant access to your complete audit history, you can’t afford to be shuffling through filing cabinets or hunting for the “latest version” of your audit checklist.
The good news? GMP audit software has evolved dramatically. What used to require complex enterprise systems costing hundreds of thousands of dollars is now accessible, cloud-based, and designed for how the quality teams actually work. But with dozens of options flooding the market, each promising to “revolutionize your quality management,” how do you separate genuine solutions from glorified spreadsheets?
This guide cuts through the marketing noise. We’ll walk you through the five audit management platforms that are actually making a difference for pharmaceutical, biotech, medical device, and food manufacturing companies. More importantly, we’ll show you exactly how to evaluate which one fits your organization, whether you’re a mid-size manufacturer running 100 audits a year or a global enterprise managing thousands across multiple continents.
By the end of this article, you’ll know:
Before diving into specific solutions, it’s important to understand what GMP audit software does and why it has become essential for modern manufacturing operations.
GMP audit software automates and digitizes the entire audit lifecycle, from planning and scheduling to execution, reporting, and corrective action tracking. These platforms replace manual, paper-based processes with centralized digital systems that enhance accuracy, accountability, and accessibility. The benefits extend beyond mere digitization. Quality management professionals report significant time savings, improved audit trail documentation, better collaboration between teams, and enhanced visibility into compliance status across multiple facilities. For organizations facing regular FDA, EMA, or other regulatory inspections, having a robust audit management system can mean the difference between smooth compliance and costly violations.
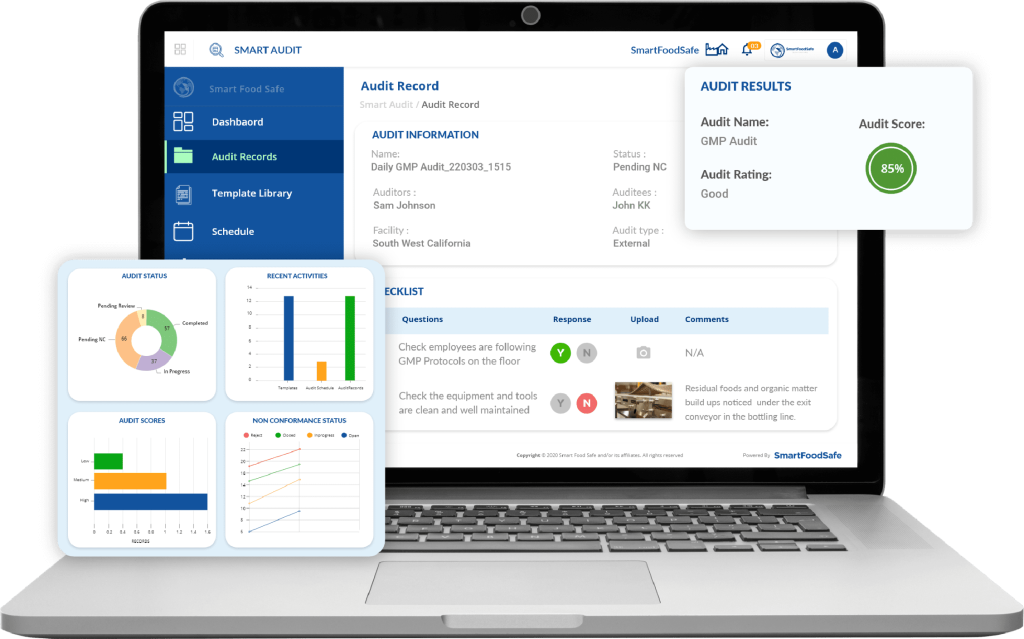
Smart Audit stands out as a comprehensive, cloud-based GMP audit management platform designed specifically for regulated industries. The solution offers an intuitive interface that makes it accessible to both quality professionals and frontline staff, reducing the learning curve that often hampers software adoption.
Key Features:

What sets Smart Audit apart is its focus on user experience and practical implementation. The platform recognizes that software is only effective if people actually use it, so it emphasizes clean design, logical workflows, and minimal clicks to complete tasks. Organizations implementing Smart Audit typically report faster time-to-value compared to more complex enterprise solutions.
The pricing model is transparent and scalable, making it suitable for mid-size manufacturers as well as large multi-site operations. Customer support receives consistently high marks, with responsive technical teams and comprehensive training resources.
Best suited for: Organizations seeking a balance between powerful functionality and user-friendly design, particularly those with multi-site operations requiring centralized audit oversight.
MasterControl has established itself as a comprehensive quality management suite that includes robust audit management capabilities. The platform serves enterprise-level organizations with complex quality ecosystems.
Key Features:
MasterControl excels in organizations that need tight integration between audit management and other quality processes. However, the complexity of the full suite can present implementation challenges, and smaller organizations may find themselves paying for functionality they don’t use.
ETQ Reliance offers a modular approach to quality management, allowing organizations to implement audit management alongside other quality modules as needed. The platform emphasizes configurability and can be tailored to specific industry requirements.
Key Features:
ETQ’s strength lies in its flexibility, though this can also mean longer implementation timelines as organizations configure the system to their specific needs. The platform requires more technical expertise to maximize its potential.
TrackWise has a long-standing reputation in the pharmaceutical industry for quality and compliance management. The audit management module integrates with their broader quality suite.
Key Features:
TrackWise is known for regulatory robustness but can feel dated in terms of user interface compared to newer cloud-native solutions. Implementation and maintenance costs tend to be higher than average.
ComplianceQuest provides a cloud-based quality management platform built on the Salesforce platform, offering audit management as part of a comprehensive quality suite.
Key Features:
The Salesforce foundation provides advantages for organizations already invested in that ecosystem but may present licensing complexity for others. The platform continues to evolve rapidly with regular feature updates.
Picking the right audit management software isn’t something you want to rush. There’s too much at stake, from compliance risks to budget commitments. Here’s how to approach this decision in a way that actually works.
Before you even look at software demos, take a hard look at your current situation. Sit down with your quality team and figure out exactly what’s broken in your current process.
Ask yourself: How many audits are we running each year? Are we talking 50 or 500? How many facilities do we need to cover, just one site or a dozen locations across different countries? Which regulatory bodies keep you up at night, FDA, EMA,or all of the above? Here’s something most vendors won’t tell you: you probably don’t need half the features they’re selling. A company doing 80 internal audits a year has very different needs than one managing 500 supplier audits across multiple continents. Figure out what’s essential for your operation before someone sells you features you’ll never use.
Also, be honest about your CAPA backlog. If you’re drowning in overdue corrective actions, you need software with strong tracking and escalation capabilities, not just fancy dashboards.
Your audit software won’t exist in a vacuum. It needs to play nice with what you already have.
Does it connect with your document management system? Can it pull data from your ERP? Will it talk to your training platform so you can see if auditors are actually qualified? These integrations matter because nobody wants to manually enter the same data into three different systems.
I’ve seen companies buy expensive software only to realize it can’t integrate with their existing systems. They end up with disconnected islands of data and frustrated quality managers doing double entry. Don’t be that company.
This is where a lot of software implementations fall apart. You can have the most powerful system in the world, but if your auditors hate using it, you’ve wasted your money.
Think about the people who’ll use this daily, not just your QA director, but the auditor walking the production floor with a tablet. Is the interface clean and straightforward, or does it require three clicks to do what should take one? Can they use it offline when they’re in areas with spotty wifi?
Here’s a pro tip: bring actual end users to the demos. Not just management. Get your auditors, your CAPA coordinators, your quality engineers in the room. Their feedback will tell you more than any sales pitch.
Sticker price isn’t the full story. Sure, one solution might have a lower monthly fee, but what about implementation? Some vendors charge $50,000 just to get you set up. Others include it.
Factor in training costs, will your vendor charge extra for each training session? What about ongoing support fees? Annual maintenance? If you need to add 20 users next year, what will that cost?
And think three to five years out. Your business will grow. Will the pricing scale reasonably, or will you get hit with massive cost jumps when you cross certain thresholds?
This should go without saying, but the software needs to meet regulatory requirements. That means 21 CFR Part 11 compliance if you’re FDA-regulated. It means proper audit trails that can’t be tampered with. It means electronic signatures that hold up under scrutiny.
Don’t just take the vendor’s word for it. Ask for their validation documentation. Ask for references from companies that have been through FDA inspections while using the software. Call those references and ask the uncomfortable questions: did the software hold up during inspection? Were there any issues with data integrity or audit trails?
What works for you today might not work in two years. Maybe you’ll acquire another facility. Maybe your audit volume will double. Maybe new regulations will require different workflows.
Will the software bend without breaking? Can you easily add users and sites? Does the vendor have a clear product roadmap, or are they still using technology from 2010?
Also, check how often they release updates. A vendor that hasn’t pushed meaningful improvements in two years probably isn’t investing in their product’s future.
You’re not just buying software, you’re entering a partnership. Some vendors disappear after the sale. Others treat implementation like an afterthought.
Ask about their implementation approach. How long does it typically take? Who owns what tasks? What happens if things go off track?
Once you’ve done your homework, don’t make this decision in a vacuum. Loop in your IT team, your operations folks, and definitely your finance department. Each brings a perspective you need.
If you can, run a pilot with your top two or three choices. Pick a real audit scenario, not some theoretical example, and see how each system handles it. You’ll learn more in one real-world test than in a dozen sales demos.
Build a simple scoring system. Weight the factors that matter most to your organization. Maybe integration capabilities are make-or-break for you. Maybe it’s mobile functionality. Whatever it is, be systematic about it so you’re making a decision based on fit, not just which salesperson you liked best.
Choosing GMP audit software is a big decision, but it doesn’t have to be overwhelming. Among the options we’ve covered, Smart Audit brings together solid functionality with the kind of user experience that actually gets adopted. But the best software is always the one that fits your specific situation. What matters most isn’t picking the platform with the longest feature list. It’s finding the one that matches how your organisation actually works, meets your regulatory requirements, and, crucially, gets used by the people who need to use it.
This isn’t just a software purchase. You’re setting up the foundation for how your organization manages quality and compliance for years to come. Take the time to get it right. Talk to vendors. Push them on the hard questions. Run pilots. Check references. And when you’re ready, start with demos that let your team get hands-on with the software. The right platform will feel intuitive pretty quickly. Your auditors will see how it makes their lives easier. Your quality managers will see the visibility they’ve been missing. That’s when you know you’ve found the right fit.
The investment you’re making today will either simplify your compliance life or become another system people work around. Choose wisely, and you’ll wonder how you ever managed audits without it.
Ready to See Smart Audit in Action?
If you’re tired of juggling spreadsheets, chasing down audit reports, and losing track of CAPAs, it might be time to see what a modern audit management system can do for your organization.
See Smart Audit for yourself with a personalized demo:
Questions about implementing audit software? Our team has helped organizations just like yours navigate this decision. Reach out at smartaudits.online and let’s talk about what would work best for your specific situation, even if that means pointing you toward a different solution.
GMP audit software is a digital platform that manages your entire audit lifecycle, from planning and scheduling to execution, reporting, and CAPA tracking. You need it if you’re still using spreadsheets or paper to manage audits, struggling to track corrective actions, or worried about what happens when regulators ask for your audit history. Modern audit software eliminates manual errors, provides instant visibility into compliance status, and ensures you’re ready for inspections at any time.
For most organizations today, cloud-based is the clear winner. You get automatic updates, lower upfront costs, anywhere-access for auditors, and you’re not maintaining servers. On-premises still makes sense if you have strict data sovereignty requirements, limited internet connectivity at facilities, or existing infrastructure you’re committed to. But cloud platforms like Smart Audit now offer enterprise-grade requirements without the IT headaches.
Most modern platforms offer integration capabilities, but the depth varies significantly. Smart Audit, for example, integrates with common document management systems, ERPs, and training platforms through APIs and standard connectors. Before committing, map out your critical integrations, what data needs to flow between systems? Ask vendors for specific examples of similar integrations they’ve completed. A platform that can’t talk to your existing systems will create more work, not less.

Have a consistent, working internal audit strategy that continuously brings about compliance in the middle of regulatory updates with Smart Audit’s audit management features.
16 Jan 2026

Your last FDA inspection didn’t go as smoothly as you’d hoped. The inspector spent an hour digging through your audit files, asking why certain CAPAs weren’t closed on time, and questioning gaps in your documentation. You had the answers, somewhere, but finding them in your maze of spreadsheets and paper trails was like searching for a needle in a haystack.
Sound familiar?
Here’s the reality: paper-based audit management and scattered Excel files aren’t just inefficient anymore, they’re a compliance liability. When regulatory bodies can show up unannounced and expect instant access to your complete audit history, you can’t afford to be shuffling through filing cabinets or hunting for the “latest version” of your audit checklist.
The good news? GMP audit software has evolved dramatically. What used to require complex enterprise systems costing hundreds of thousands of dollars is now accessible, cloud-based, and designed for how the quality teams actually work. But with dozens of options flooding the market, each promising to “revolutionize your quality management,” how do you separate genuine solutions from glorified spreadsheets?
This guide cuts through the marketing noise. We’ll walk you through the five audit management platforms that are actually making a difference for pharmaceutical, biotech, medical device, and food manufacturing companies. More importantly, we’ll show you exactly how to evaluate which one fits your organization, whether you’re a mid-size manufacturer running 100 audits a year or a global enterprise managing thousands across multiple continents.
By the end of this article, you’ll know:
Before diving into specific solutions, it’s important to understand what GMP audit software does and why it has become essential for modern manufacturing operations.
GMP audit software automates and digitizes the entire audit lifecycle, from planning and scheduling to execution, reporting, and corrective action tracking. These platforms replace manual, paper-based processes with centralized digital systems that enhance accuracy, accountability, and accessibility. The benefits extend beyond mere digitization. Quality management professionals report significant time savings, improved audit trail documentation, better collaboration between teams, and enhanced visibility into compliance status across multiple facilities. For organizations facing regular FDA, EMA, or other regulatory inspections, having a robust audit management system can mean the difference between smooth compliance and costly violations.
Smart Audit stands out as a comprehensive, cloud-based GMP audit management platform designed specifically for regulated industries. The solution offers an intuitive interface that makes it accessible to both quality professionals and frontline staff, reducing the learning curve that often hampers software adoption.
Key Features:

What sets Smart Audit apart is its focus on user experience and practical implementation. The platform recognizes that software is only effective if people actually use it, so it emphasizes clean design, logical workflows, and minimal clicks to complete tasks. Organizations implementing Smart Audit typically report faster time-to-value compared to more complex enterprise solutions.
The pricing model is transparent and scalable, making it suitable for mid-size manufacturers as well as large multi-site operations. Customer support receives consistently high marks, with responsive technical teams and comprehensive training resources.
Best suited for: Organizations seeking a balance between powerful functionality and user-friendly design, particularly those with multi-site operations requiring centralized audit oversight.
MasterControl has established itself as a comprehensive quality management suite that includes robust audit management capabilities. The platform serves enterprise-level organizations with complex quality ecosystems.
Key Features:
MasterControl excels in organizations that need tight integration between audit management and other quality processes. However, the complexity of the full suite can present implementation challenges, and smaller organizations may find themselves paying for functionality they don’t use.
ETQ Reliance offers a modular approach to quality management, allowing organizations to implement audit management alongside other quality modules as needed. The platform emphasizes configurability and can be tailored to specific industry requirements.
Key Features:
ETQ’s strength lies in its flexibility, though this can also mean longer implementation timelines as organizations configure the system to their specific needs. The platform requires more technical expertise to maximize its potential.
TrackWise has a long-standing reputation in the pharmaceutical industry for quality and compliance management. The audit management module integrates with their broader quality suite.
Key Features:
TrackWise is known for regulatory robustness but can feel dated in terms of user interface compared to newer cloud-native solutions. Implementation and maintenance costs tend to be higher than average.
ComplianceQuest provides a cloud-based quality management platform built on the Salesforce platform, offering audit management as part of a comprehensive quality suite.
Key Features:
The Salesforce foundation provides advantages for organizations already invested in that ecosystem but may present licensing complexity for others. The platform continues to evolve rapidly with regular feature updates.
Picking the right audit management software isn’t something you want to rush. There’s too much at stake, from compliance risks to budget commitments. Here’s how to approach this decision in a way that actually works.
Before you even look at software demos, take a hard look at your current situation. Sit down with your quality team and figure out exactly what’s broken in your current process.
Ask yourself: How many audits are we running each year? Are we talking 50 or 500? How many facilities do we need to cover, just one site or a dozen locations across different countries? Which regulatory bodies keep you up at night, FDA, EMA,or all of the above? Here’s something most vendors won’t tell you: you probably don’t need half the features they’re selling. A company doing 80 internal audits a year has very different needs than one managing 500 supplier audits across multiple continents. Figure out what’s essential for your operation before someone sells you features you’ll never use.
Also, be honest about your CAPA backlog. If you’re drowning in overdue corrective actions, you need software with strong tracking and escalation capabilities, not just fancy dashboards.
Your audit software won’t exist in a vacuum. It needs to play nice with what you already have.
Does it connect with your document management system? Can it pull data from your ERP? Will it talk to your training platform so you can see if auditors are actually qualified? These integrations matter because nobody wants to manually enter the same data into three different systems.
I’ve seen companies buy expensive software only to realize it can’t integrate with their existing systems. They end up with disconnected islands of data and frustrated quality managers doing double entry. Don’t be that company.
This is where a lot of software implementations fall apart. You can have the most powerful system in the world, but if your auditors hate using it, you’ve wasted your money.
Think about the people who’ll use this daily, not just your QA director, but the auditor walking the production floor with a tablet. Is the interface clean and straightforward, or does it require three clicks to do what should take one? Can they use it offline when they’re in areas with spotty wifi?
Here’s a pro tip: bring actual end users to the demos. Not just management. Get your auditors, your CAPA coordinators, your quality engineers in the room. Their feedback will tell you more than any sales pitch.
Sticker price isn’t the full story. Sure, one solution might have a lower monthly fee, but what about implementation? Some vendors charge $50,000 just to get you set up. Others include it.
Factor in training costs, will your vendor charge extra for each training session? What about ongoing support fees? Annual maintenance? If you need to add 20 users next year, what will that cost?
And think three to five years out. Your business will grow. Will the pricing scale reasonably, or will you get hit with massive cost jumps when you cross certain thresholds?
This should go without saying, but the software needs to meet regulatory requirements. That means 21 CFR Part 11 compliance if you’re FDA-regulated. It means proper audit trails that can’t be tampered with. It means electronic signatures that hold up under scrutiny.
Don’t just take the vendor’s word for it. Ask for their validation documentation. Ask for references from companies that have been through FDA inspections while using the software. Call those references and ask the uncomfortable questions: did the software hold up during inspection? Were there any issues with data integrity or audit trails?
What works for you today might not work in two years. Maybe you’ll acquire another facility. Maybe your audit volume will double. Maybe new regulations will require different workflows.
Will the software bend without breaking? Can you easily add users and sites? Does the vendor have a clear product roadmap, or are they still using technology from 2010?
Also, check how often they release updates. A vendor that hasn’t pushed meaningful improvements in two years probably isn’t investing in their product’s future.
You’re not just buying software, you’re entering a partnership. Some vendors disappear after the sale. Others treat implementation like an afterthought.
Ask about their implementation approach. How long does it typically take? Who owns what tasks? What happens if things go off track?
Once you’ve done your homework, don’t make this decision in a vacuum. Loop in your IT team, your operations folks, and definitely your finance department. Each brings a perspective you need.
If you can, run a pilot with your top two or three choices. Pick a real audit scenario, not some theoretical example, and see how each system handles it. You’ll learn more in one real-world test than in a dozen sales demos.
Build a simple scoring system. Weight the factors that matter most to your organization. Maybe integration capabilities are make-or-break for you. Maybe it’s mobile functionality. Whatever it is, be systematic about it so you’re making a decision based on fit, not just which salesperson you liked best.
Choosing GMP audit software is a big decision, but it doesn’t have to be overwhelming. Among the options we’ve covered, Smart Audit brings together solid functionality with the kind of user experience that actually gets adopted. But the best software is always the one that fits your specific situation. What matters most isn’t picking the platform with the longest feature list. It’s finding the one that matches how your organisation actually works, meets your regulatory requirements, and, crucially, gets used by the people who need to use it.
This isn’t just a software purchase. You’re setting up the foundation for how your organization manages quality and compliance for years to come. Take the time to get it right. Talk to vendors. Push them on the hard questions. Run pilots. Check references. And when you’re ready, start with demos that let your team get hands-on with the software. The right platform will feel intuitive pretty quickly. Your auditors will see how it makes their lives easier. Your quality managers will see the visibility they’ve been missing. That’s when you know you’ve found the right fit.
The investment you’re making today will either simplify your compliance life or become another system people work around. Choose wisely, and you’ll wonder how you ever managed audits without it.
Ready to See Smart Audit in Action?
If you’re tired of juggling spreadsheets, chasing down audit reports, and losing track of CAPAs, it might be time to see what a modern audit management system can do for your organization.
See Smart Audit for yourself with a personalized demo:
Questions about implementing audit software? Our team has helped organizations just like yours navigate this decision. Reach out at smartaudits.online and let’s talk about what would work best for your specific situation, even if that means pointing you toward a different solution.
GMP audit software is a digital platform that manages your entire audit lifecycle, from planning and scheduling to execution, reporting, and CAPA tracking. You need it if you’re still using spreadsheets or paper to manage audits, struggling to track corrective actions, or worried about what happens when regulators ask for your audit history. Modern audit software eliminates manual errors, provides instant visibility into compliance status, and ensures you’re ready for inspections at any time.
For most organizations today, cloud-based is the clear winner. You get automatic updates, lower upfront costs, anywhere-access for auditors, and you’re not maintaining servers. On-premises still makes sense if you have strict data sovereignty requirements, limited internet connectivity at facilities, or existing infrastructure you’re committed to. But cloud platforms like Smart Audit now offer enterprise-grade requirements without the IT headaches.
Most modern platforms offer integration capabilities, but the depth varies significantly. Smart Audit, for example, integrates with common document management systems, ERPs, and training platforms through APIs and standard connectors. Before committing, map out your critical integrations, what data needs to flow between systems? Ask vendors for specific examples of similar integrations they’ve completed. A platform that can’t talk to your existing systems will create more work, not less.

Have a consistent, working internal audit strategy that continuously brings about compliance in the middle of regulatory updates with Smart Audit’s audit management features.
Have a consistent, working internal audit strategy that continuously brings about compliance in the middle of regulatory updates with Smart Audit’s audit management features.
Signup to receive latest news, insights and updates on audits management